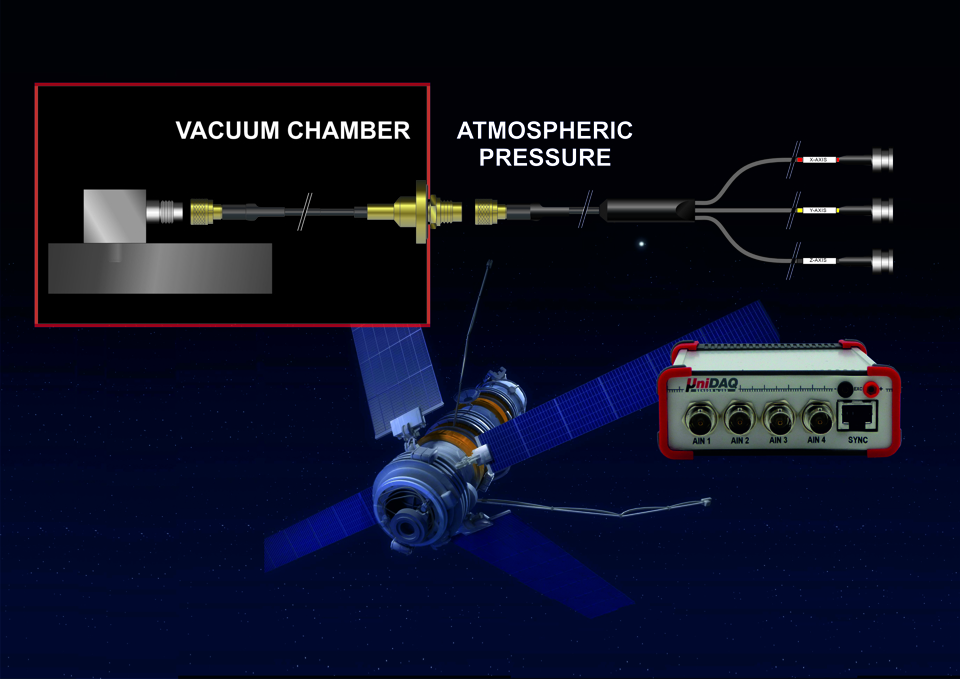
- +++ kompakte DAQ/Datenlogger (≈ 12x11x4 cm ) für << 2000 EUR! Preiswert aber hochwertig! +++
- Unser Sensorlexikon: Wissen über Sensorik
- Smart Sensors: CAN-basierende Machinendiagnose: CAN-MD®
- µSTICK IEPE-V für IEPE Sensoren: In-Line Modul für Spannung- statt Stromversorgung
- Innovativ: IEPE Sensoren bis 200°C: Dytran 3525A-Serie
- Auslaufmodell: Kraftsensor ELFF nur 149,- EUR
- Lesenswert: Die 30 Artikel der Allgemeinen Erklärung der Menschenrechte, Resolution 217 der UN-Vollversammlung vom 10. Dezember 1948, SItzung 183
- cookiefreie Homepage: Wir sammeln oder speichern keine Daten von Ihnen!
Mach messbar, was noch nicht messbar ist. — Galileo Galilei
Mit Sensoren werden somit physikalische Größen messbar gemacht. Die Sensorsignale werden mit Elektronik zur Signalverarbeitung in nutzbare elektrische, manchmal auch optische Signale gewandelt.
Mit Hilfe von Datenerfassungssystemen oder Datenloggern werden die optimierten Signale aufgezeichnet und analysiert. Mit intelligenter Sensorik geschieht die Signalverarbeitung und Analyse im Sensor.
Beispiele für Sensoren sind: Beschleunigungssensoren, Drucksensoren, Kraftsensoren, Drehmomentsensoren, Wegsensoren oder Neigungssensoren.
Seit der Gründung im Jahr 1995, bieten wir kundenorientierte Lösungen für Sensor- und Messtechnikaufgaben an. Der Name disynet sowie unser Motto „Made to Measure“ stehen für maßgeschneiderte Konzepte für die einzigartigen Anwendungen unserer Kunden. Sie sind für uns Verpflichtung und Kapital in einem.
Unser Produktspektrum umfasst eine Vielzahl an Messwertaufnehmern und Messtechniksystemen für unterschiedliche Messgrößen mit jeweils verschiedenen, zugrundeliegenden Sensortechnologien. Unsere Produkte werden hauptsächlich in den Bereichen „Test & Measurement“, Prüfstandsmessung, Forschung und Entwicklung sowie Zustandsüberwachung eingesetzt, unter anderem für die Automobilindustrie, Luft- und Raumfahrt, Schiene und Bahn, Windkraftanlagen, Landmaschinen, Bauwerke, usw.
Unsere Homepage zeigt davon nur eine kleine Auswahl an Sensorik.

Wussten Sie schon…?
Sie finden uns auch auf:
Die disynet GmbH ist bereits seit über 25 Jahren Ihr kompetenter Ansprechpartner in Sachen Sensoren & Messtechnik!